Indoor Air Quality Testing: Oxygen Levels and Oxygen Deprivation Effects

Oxygen Atomic Structure – Oxygen Levels in Indoor Air Quality (IAQ)
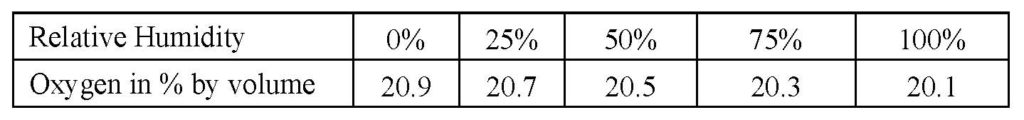
One metric that is commonly overlooked in air quality studies is the oxygen percentage in the indoor environment with respect to normal atmospheric composition and how sensitive the human body is to this level. The oxygen levels on present day 21st Century Earth are nominally 20.9 % IF the relative humidity is 0%. (only found in extremely arid regions or dehumidified rooms such as an attic in summer) At one time in the distant past (the days of the dinosaurs) the oxygen levels on Earth were as high as 35 %.
The water vapor in the air displaces oxygen to some extent, so the more humid the air, the lower the % of oxygen. Here is a chart for reference:
The body (particularly the brain) requires oxygen to function because it is a critical component in the cellular respiration process. (also known as the Krebs or Citric Acid Cycle) There are 42 steps in this cycle, and oxygen comes in at the last stage to combine with the H+ hydrogen proton gradient built up in the mitochondria (the “power plant” which is the main producer of ATP – also known as adenosine triphosphate which is the “energy currency” of biological organisms) of cells. Oxygen combined with hydrogen forms water which is the desired byproduct. Without it, the hydrogen atoms would drive the pH balance to a dangerously acidic level and denature proteins and kill all cellular functions.
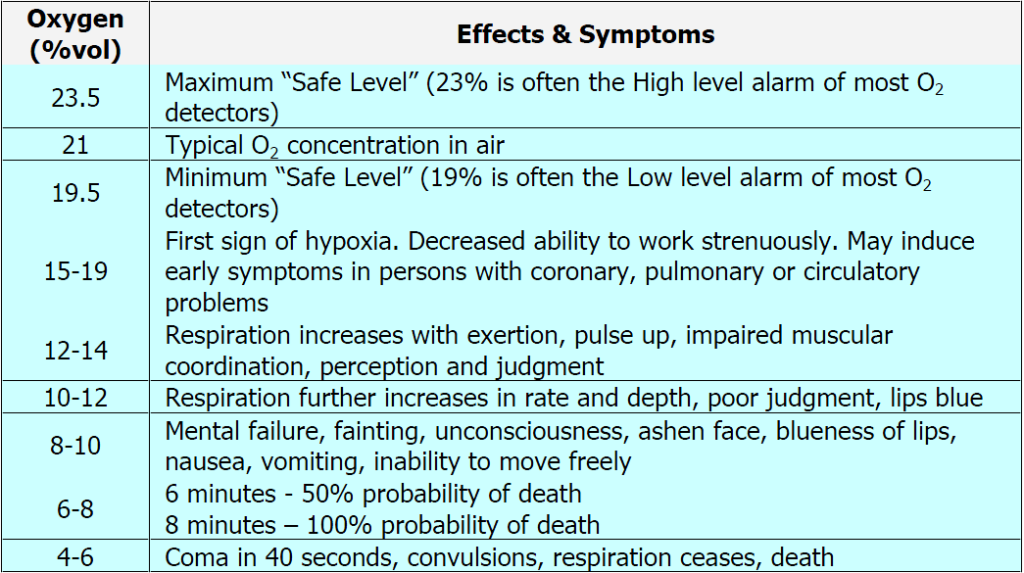
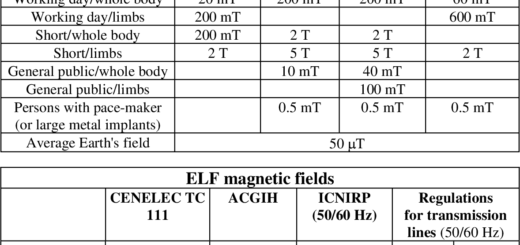
Notice that the safe range of oxygen is from 19.5 – 23.5 % which is only a 4 point percentage window. This illustrates how narrow the levels are between too much and too little oxygen. Too much oxygen (oxygen toxicity) can slow breathing levels to a rate that does not displace enough carbon dioxide (CO2) – a condition known as carbon dioxide narcosis. This typically only occurs when too much oxygen from a supplemental system such as tanks used in scuba diving or medical breathing equipment that is not adjusted correctly. This is virtually never an indoor air quality issue, but it is mentioned here to illustrate that even chemicals typically considered harmless or helpful can have dangerous effects at high enough levels.
So why would oxygen deprivation be an issue – aren’t oxygen levels homogeneous everywhere?
Not necessarily.
Areas with more forestation and foliage typically have higher levels of oxygen than outlying scrub prairies, but more to the point, deep urban environments often suffer from a shortage of oxygen for several reasons:
- Lack of oxygen emitting plants and trees
- High consumption of oxygen by dense populations such as found in heavily urbanized cites like Houston, Austin, the Dallas / Fort Worth region, etc. vehicles and other combustion motors
- Oxidation / formation of chemicals that bind up oxygen molecules such as sulfur dioxides and nitrogen oxides
- Displacement of oxygen by other gases and vapors such as water vapor, carbon dioxide, etc.
This is why particularly in downtown environments or near other heavily trafficked / polluted areas it is a good idea to know how much oxygen is being received as the chart above demonstrates that even a drop of 1 % or so (say 20.1% to 19.1%) can induce the subtle but deleterious effects of hypoxia.
Too much oxygen known as hyperoxia or “oxygen toxicity” causes damage depending on oxygen %, individual lung capacity, and exposure time. Part of the problem is that with too much oxygen, the body is unable to get rid of carbon dioxide effectively. Here is an article that goes into more medical detail:
https://www.ncbi.nlm.nih.gov/books/NBK430743/
To quote the early physician / alchemist Paracelsus: ” Poison is in everything, and no thing is without poison. The dosage makes it either a poison or a remedy.”
Questions from Readers about Oxygen Levels
Is there a home wall mounted O2 measuring device that exist on the retail market. Something like my home thermostat ? If yes, can someone point me in the right direction. Most inquiries I type into ‘ Google Search’ gives me a personal fingertip O2 sensor that measures Oxygenated Blood Levels. – Not what I am interested in. Thank you for you response.
Jeff Elliott
Houston, TX
Jeff,
>Is there a home wall mounted O2 measuring device that exist on the retail market. Something like my >home thermostat ?
Good question!
>Most inquiries I type into ‘ Google Search’ gives me a personal fingertip O2 sensor that measures >Oxygenated Blood Levels. – Not what I am interested in. Thank you for you response.
Yes, those fingertip monitors are known as pulse oximeters (also known as plethysmographs) which I personally recommend for a first aid kit IF you are qualified to select and use one. (medically trained)
What you are asking about in regard to Oxygen (O2) sensors that are mounted on the wall are typically only found at the commercial level – often in the configuration of what is known as a 4 gas analyzer / multi-gas analyzer which measures oxygen, carbon monoxide and generally combustible gases which are measured in LEL %. (Lower Explosive Level)
Because I only use field instrumentation for measurement and not a permanent / semi-permanent unit, I cannot give an official recommendation for one. Just use the search terms mentioned above and you may find a qualified device recommended by a professional as these are used in many industries.
As an aside, you will probably have an easier (and less expensive) path in purchasing a meter that measures carbon DIOXIDE (not monoxide) or CO2 meter in order to gauge your ventilation quality. While at this writing I cannot give a direct numerical correlation between the CO2 and O2 balance in an environment, it makes sense that in general if your ventilation is good, the CO2 will be decreased with a somewhat correlated increase in oxygen. (but we are talking about fractions of a percent) Reference the table above showing humidity correlation with oxygen under relatively ideal conditions.
As a side safety note, it is a good idea to have a reliable, accurate carbon monoxide detector to provide early warning of faulty water heaters, central gas heating, or an idling car in a garage which is near a living area. The monoxide levels in a closed garage will climb to dangerous levels in a very short time with virtually any combustion engine. Propane forklifts which are malfunctioning, improperly tuned, etc. are hazards in warehouse environments.
– JAG of ScanTech
WHO IS A CERTIFIED EMF CONSULTANT NEAR ME?
ScanTech Technical Consulting is owned and operated by Joel-Anthony Gray who has over 22 years of experience as an EMF consultant, EMI troubleshooting expert and nuclear radiation testing consultant for a variety of commercial and industrial customers. While there is currently no such thing as a Certified EMF Consultant as far as the United States industrial and scientific community at large is concerned, what ScanTech offers is extensive education, experience and multiple certifications in non-ionizing radiation, industrial safety and hygiene. (listed here EMF Credentials) He holds numerous degrees and certifications which qualify him for this level of expertise. Call 214.912.4691
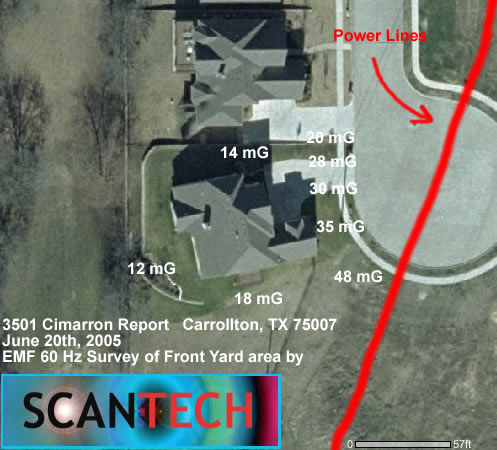
WHO DOES ELECTROMAGNETIC FIELD TESTING NEAR ME?
ScanTech Technical Consulting professional commercial electromagnetic field testing and radio frequency testing in the Dallas – Fort Worth Texas area to detect and measure exposures and health risks (if any) to electromagnetic fields and radiation. Call 214.912.4691
WHO OR WHAT COMPANY DOES EMF TESTING NEAR ME?
ScanTech Technical Consulting professional commercial electromagnetic field testing and radio frequency testing in the Dallas – Fort Worth Texas area to detect and measure exposures and health risks (if any) to electromagnetic fields and radiation. Call 214.912.4691
WHO OR WHAT COMPANY DOES RADIO FREQUENCY (RF) & CELLULAR RADIATION (5G) TESTING NEAR ME?
ScanTech Technical Consulting provides commercial radio frequency (RF) testing in the Dallas – Fort Worth Texas area to detect and measure exposures to all forms and frequencies of radio frequency radiation. Call 214.912.4691
WHO OR WHAT COMPANY DOES PACEMAKER EMI TESTING AND ELECTRICAL INTERFERENCE SURVEYS FOR IMPLANTED BIOMEDICAL DEVICES NEAR ME?
ScanTech Technical Consulting provides professional EMI testing for pacemakers and other biomedical implants throughout the United States to detect and measure exposures to all forms of interference from electromagnetic radiation. Call 214.912.4691
WHO OR WHAT COMPANY DOES RF RADIO FREQUENCY CELL TOWER TESTING NEAR ME?
ScanTech Technical Consulting provides commercial cellular testing in the Dallas – Fort Worth Texas area to detect and measure exposures to all forms of RF. Call 214.912.4691
WHO OR WHAT COMPANY DOES ELECTROMAGNETIC RADIATION TESTING NEAR ME?
ScanTech Technical Consulting provides commercial radio frequency (RF) testing in the Dallas – Fort Worth Texas area to detect and measure exposures to all forms of electromagnetic radiation. Call 214.912.4691
WHO OR WHAT COMPANY DOES ELECTRICAL INTERFERENCE TESTING NEAR ME?
ScanTech Technical Consulting provides professional radio frequency (RF) testing in the Dallas – Fort Worth Texas area to detect and measure exposures to all forms of electromagnetic radiation. Call 214.912.4691
WHO OR WHAT COMPANY DOES NUCLEAR RADIOACTIVITY AND RADIATION TESTING NEAR ME?
ScanTech Technical Consulting provides professional radioactivity testing in the Dallas – Fort Worth, Houston, Austin and San Antonio areas of Texas to detect and measure exposure to ionizing, nuclear or atomic radiation. Call 214.912.4691
WHO OR WHAT COMPANY DOES DALLAS AREA PHOTOMETRIC LIGHTING SURVEYS OR EXTERIOR LIGHTING STUDIES NEAR ME?
ScanTech Technical Consulting provides professional photometric testing in the Dallas Texas area for commercial clients to help pass lighting ordinances for their Certificate of Occupancy. Call 214.912.4691
WHO OR WHAT COMPANY DOES ESD / ELECTROSTATIC DISCHARGE TESTING NEAR ME?
ScanTech Technical Consulting provides ESD testing in the Dallas – Fort Worth, Austin, San Antonio and Houston Texas area to evaluate, measure and perform Electrostatic Discharge testing and consulting for sensitive environments. Call 214.912.4691
We often serve clients in Texas, Michigan and Iowa. Cities for onsite commercial testing and inspection services include: Plano, Highland Park, University Park, Park Cities, Las Colinas, Arlington, Fort Worth, Houston, Austin, San Antonio, Shreveport, Grapevine, Frisco, Denton, McKinney, Allen, Lewisville, Irving, Mesquite, Bedford, Euless, Richardson, Coppell, Grand Prairie, Garland, Addison, Farmers Branch, Rockwall, Carrollton, Parker, Rowlett, Lucas, Fairview, Park Cities, Keller, Roanoke, The Colony, Highland Village, Lake Dallas, Corinth, Prosper, Duncanville, Lancaster, Rowlett, Royse City, Princeton, Trophy Club, Southlake, Hurst, Round Rock, Georgetown, San Marcos, Cedar Park, The Woodlands and Spring. Counties served include Dallas, Collin, Denton, Tarrant, Rockwall, Harris and Travis County.
LARGER COMMERCIAL PROJECT SERVICE RANGE – NATIONAL & INTERNATIONAL
Alabama | Alaska | Arizona | Arkansas | California | Colorado | Connecticut | Delaware | Florida | Georgia | Hawaii | Idaho | Illinois | Indiana | Iowa | Kansas | Kentucky | Louisiana | Maine | Maryland | Massachusetts | Michigan | Minnesota | Mississippi | Missouri | Montana | Nebraska | Nevada | New Hampshire | New Jersey | New Mexico | New York | North Carolina | North Dakota | Ohio | Oklahoma | Oregon | Pennsylvania | Rhode Island | South Carolina | South Dakota | Tennessee | Texas | Utah | Vermont | Virginia | Washington | West Virginia | Wisconsin | Wyoming | Washington D.C. (District of Columbia)
Major US Cities: Chicago, Detroit, Phoenix, Denver, Salt Lake City, Miami, Grand Rapids, Lansing, Nashville, Memphis, Atlanta, Charleston, Raleigh-Durham, Charlotte, Des Moines, Milwaukee
Countries served include the United States, Canada, Australia, New Zealand the UK / United Kingdom (England, Scotland, Wales, Ireland) and Western Europe.
*LEGAL NOTICE*
All information on this website either written or implied is the express opinion of ScanTech Technical Consulting. ScanTech Technical Consulting and it’s owners are not responsible or liable for any damages arising from the misuse, misinterpretation or other consequences of the content of this website either in part or in whole. This includes all external weblinks, PDF documents, photos or other references (informational or otherwise) to 3rd parties including government agencies, health organizations, etc.
15770 Dallas Pkwy Suite # 900 Dallas, TX 75248 (Not Accepting Visitors due to C19)
Phone: (214).912.4691 https://emfsurvey.com
ScanTech Technical Consulting: Professional Electromagnetic EMF Consulting / EMI Testing for Biomedical Implant & Equipment, Indoor Air Quality (IAQ) Testing & Environmental Studies and Inspections
COPYRIGHT 2002 – 2024
























Is there a home wall mounted O2 measuring device that exist on the retail market. Something like my home thermostat ? If yes, can someone point me in the right direction. Most inquiries I type into ‘ Google Search’ gives me a personal fingertip O2 sensor that measures Oxygenated Blood Levels. – Not what I am interested in. Thank you for you response.
Jeff Elliott
Houston, TX
Jeff,
>Is there a home wall mounted O2 measuring device that exist on the retail market. Something like my >home thermostat ?
Good question!
>Most inquiries I type into ‘ Google Search’ gives me a personal fingertip O2 sensor that measures >Oxygenated Blood Levels. – Not what I am interested in. Thank you for you response.
Yes, those fingertip monitors are known as pulse oximeters which I personally recommend for a first aid kit IF you are qualified to select and use one. (medically trained)
What you are asking about in regard to Oxygen (O2) sensors that are mounted on the wall are typically only found at the commercial level – often in the configuration of what is known as a 4 gas analyzer / multi-gas analyzer which measures oxygen, carbon monoxide and generally combustible gases which are measured in LEL %. (Lower Explosive Level)
Because I only use field instrumentation for measurement and not a permanent / semi-permanent unit, I cannot give an official recommendation for one. Just use the search terms mentioned above and you may find a qualified device recommended by a professional as these are used in many industries.
As an aside, you will probably have an easier (and less expensive) path in purchasing a meter that measures carbon DIOXIDE (not monoxide) or CO2 meter in order to gauge your ventilation quality. While at this writing I cannot give a direct numerical correlation between the CO2 and O2 balance in an environment, it makes sense that in general if your ventilation is good, the CO2 will be decreased with a somewhat correlated increase in oxygen. (but we are talking about fractions of a percent) Reference the table above showing humidity correlation with oxygen under relatively ideal conditions.
As a side safety note, it is a good idea to have a reliable, accurate carbon monoxide detector to provide early warning of faulty water heaters, central gas heating, or an idling car in a garage which is near a living area. The monoxide levels in a closed garage will climb to dangerous levels in a very short time with virtually any combustion engine. Propane forklifts which are malfunctioning, improperly tuned, etc. are hazards in warehouse environments.
-JAG of ScanTech